Our Research
Our research group focuses on the design and development of novel therapeutic agents through the integration of medicinal chemistry, chemical biology, and advanced computational drug discovery approaches. We work on various projects targeting different disease areas, with a particular emphasis on cancer and infectious diseases.
Antimicrobial Drug Development
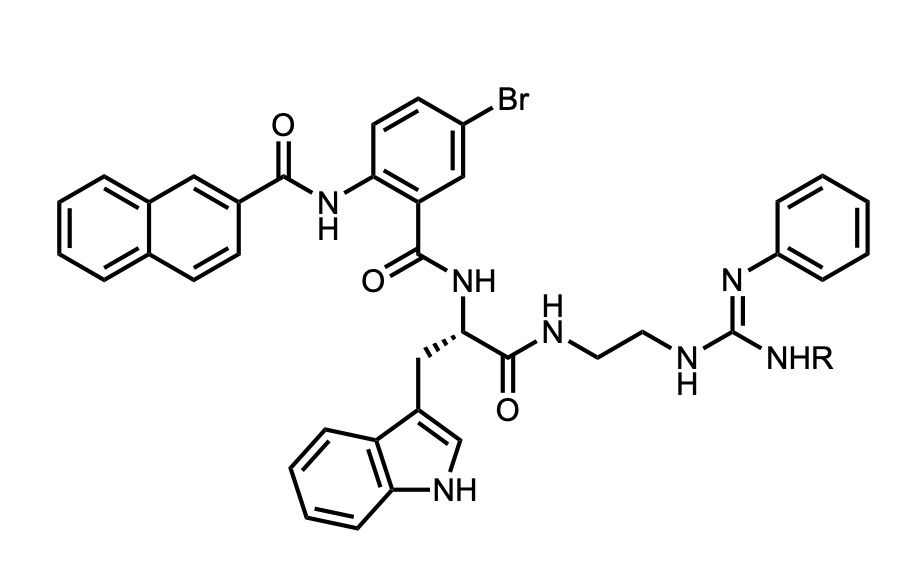
1. Antimicrobial Peptide Mimics
Our group is developing novel antimicrobial peptide mimics to combat drug-resistant bacteria. This research includes:
- Design and synthesis of peptide mimics
- Structure-activity relationship studies
- Mechanism of action studies
- In vitro and in vivo evaluation
Key Publications
- Ghayah Bahatheg, Rajesh Kuppusamy, Lissy Hartmann, Charles Gordon Cranfield, David StClair Black, Mark D. P. Willcox, Naresh Kumar. Antibacterial peptidomimetics based on guanidine-functionalized di-tertiary amides. RSC Medicinal Chemistry 2025. DOI: 10.1039/D5MD00688K
- Kuppusamy R, Yasir M, Yu TT, Voli F, Vittorio O, Miller MJ, Lewis P, Black DS, Willcox M, Kumar N. Tuning the anthranilamide peptidomimetic design to selectively target planktonic bacteria and biofilm. Antibiotics 2023, 12, 585. DOI: 10.3390/antibiotics12030585
2. Antimicrobial Hydrogel
We are investigating antimicrobial peptide-mimics that can also form hydrogels. This work predominantly focuses on:
- Determining the structure-functional properties of novel low molecular weight (LMW) hydrogelating antimicrobial agents
- Incorporating nitric-oxide releasing functional groups to form dual-action antimicrobial hydrogels
- Development of "smart" controllable photoswitchable hydrogels with antimicrobial properties
Key Publications
- Attard, S.T.; Aldilla, V.R.; Kuppusamy, R.; Chen, R.; Black, D.S.; Thordarson, P.; Willcox, M.D.P.; Kumar, N. Tuning the Structure–Functional Properties Within Peptide-Mimicking Antimicrobial Hydrogels. Antibiotics 2025, 14, 1118. DOI: 10.3390/antibiotics14111118
- Aldilla, V.R., Chen, R., Kuppusamy, R. et al. Hydrogels with intrinsic antibacterial activity prepared from naphthyl anthranilamide (NaA) capped peptide mimics. Sci Rep 12, 22259 (2022). DOI: 10.1038/s41598-022-26426-1
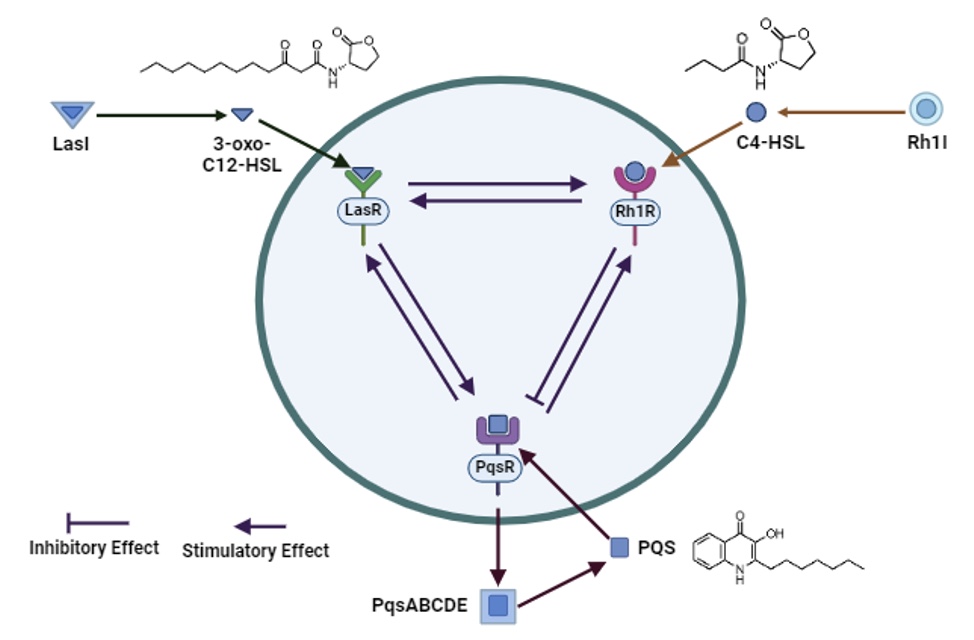
3. Quorum Sensing Inhibitors
The emergence of multi-drug resistance in common human pathogens has highlighted the need to develop novel classes of antimicrobials for the treatment of human disease. A number of projects are available in this area focussing on a combination of organic synthesis, molecular modelling, and in vitro and in vivo antimicrobial screening. This project will develop novel antagonists of bacterial signalling pathways, which inhibit the regulatory quorum sensing communication pathways of bacteria, and will model the receptor-ligand interaction using the X-ray crystal structures of bacterial signal receptors e.g. Pseudomonas quinolone system (PQS).
Key Publications
- Kosana Sai Chaitanya, Tsz Tin Yu, Hrushikesh Chaudhari, Nidhi Orekonday, Naresh Kumar, Ruchi Jain Dey, Sankaranarayanan Murugesan, Kondapalli Venkata Gowri Chandra Sekhar. Design, Synthesis and Evaluation of Indole-Based 1,2,3-Triazoles as Potential Quorum Sensing and Biofilm Inhibitors Against Pseudomonas aeruginosa. Archiv der Pharmazie 2026, 359, e70209. DOI: 10.1002/ardp.70209
- Suresh D, Yu TT, Kuppusamy R, Sabir S, Das T, Black DStC, Willcox M, Kumar N. Novel cationic dihydropyrrol-2-one compounds as antimicrobial agents and quorum sensing inhibitors. Bioorg. Med. Chem. 2025, 122, 118137. DOI
4. Inhibitors of Bacterial Transcription Initiation
The enzyme RNA polymerase (RNAP) that transcribes DNA into RNA is highly conserved across species. However, the factors that regulate the activity of RNAP are target-specific. Therefore, the unique interaction of sigma factors with RNAP in bacteria represents an ideal target for the development of small molecules that can specifically inhibit this interaction3. In this project new molecules that target these essential protein-protein interactions will be rationally designed and synthesized, and evaluated for their antimicrobial efficacy. These new small molecules would represent lead compounds for the development of new antibiotics.

Key Publications
- Kumar N et al. Small molecule inhibitors of bacterial transcription complex formation. Bioorganic and Medicinal Chemistry Letters 2017. DOI: 10.1016/j.bmcl.2017.08.036
- Mielczarek M, Thomas RV, Ma C, Kandemir H, Yang X, Bhadbhade M, Black DStC, Griffith R, Lewis P, Kumar N. Synthesis and biological activity of novel mono-indole and mono-benzofuran inhibitors of bacterial transcription initiation complex formation. Bioorganic & Medicinal Chemistry 2015, 23(8):1763–1775. DOI: 10.1016/j.bmc.2015.02.037
5. Natural Products as Antibacterial Agents
The rise of antimicrobial resistance highlights the urgent need for new antibacterial agents. This project investigates marine sponges as a source of structurally unique natural products with potential antibacterial activity. Bioactive compounds are isolated from an Australian Aplysina sp. sponge using chromatographic techniques and identified through spectroscopic analysis (NMR and HRES-MS). In addition, synthetic analogues are prepared to explore the development of new antibacterial leads.

Cancer Drug Discovery
1. Targeting MYCN with New Scaffolds for Anticancer Discovery
Neuroblastoma (NB) and glioblastoma (GBM) are aggressive cancers with poor prognosis, often driven by MYCN overexpression. MYCN is challenging to target directly due to its nuclear localization and lack of druggable pockets. Its stability is regulated by ubiquitination, and de-ubiquitinating enzymes (DUBs) like USP5 prevent MYCN degradation. Inhibition of USP5 restores p53-mediated cell cycle control and induces apoptosis. This project aims to design and synthesize novel scaffolds to inhibit USP5, evaluating their anticancer efficacy in both NB and GBM models.
Key Publications
- Gadde S, Leung YC, Bhadbade M, Cheung BB, Black DS, Kumar N (2020) Synthesis of a novel library of 1-substituted pyrido [1, 2-a] benzimidazoles. Australian Journal of Chemistry 73: 1208-1218. DOI: 10.1071/CH20173
- Gadde S, Kleynhans A, Holien JK, Bhadbhade M, Nguyen PLD, Mittra R, Yu TT, Carter DR, Parker MW, Marshall GM, Cheung BB, Kumar N. Pyrimido[1,2-a]benzimidazoles as inhibitors of oncoproteins ubiquitin specific protease 5 and MYCN in the childhood cancer neuroblastoma. Bioorganic Chemistry 2023, 136, 106462. DOI: 10.1016/j.bioorg.2023.106462
2. Design And Synthesis of Novel Inhibitors Targeting Acute Myeloid Leukemia
(in collaboration with Dr Daniel Wenholz UNSW) Acute Myeloid Leukemia (AML) is a blood cancer (leukaemia) that represents ~40% of all new adult-onset leukemias in Australia. It is characterised by the overproduction of abnormal myeloblasts in the bone marrow, preventing healthy myeloblast, platelet and erythrocyte production. FMS-like tyrosine kinase 3 (FLT3) is a class III tyrosine kinase receptor involved in the regulation of hematopoietic cell differentiation, survival and proliferation. FLT3 mutations are among the most frequently identified mutations involved in leukaemia development and occur in approximately 28% of AML patients. Mutations of FLT3 have been associated with a poor prognosis, specifically adverse disease features, poor survival and a reduced rate of remission. A derivative of phenoxodiol has been identified as a screening hit compound for inhibition against FLT3 mutants. The overall aim of this project is to investigate the structure activity relationship on FLT3 through the synthesis of a library of novel analogues.
3. Novel Heterocyclic Analogues of Isoflavones as Anticancer Drug
The isoflavones are the largest and most widely studied class of phytoestrogens displaying potent and selective cytotoxicity against cancer cells, with low toxicity to healthy cells. During the past five years we have developed several new phenoxodiol conjugates with potent biological activities, and have also incorporated phenoxodiol in a cyclodextrin formulation, which shows increased aqueous solubility. Furthermore we have covalently conjugated phenoxodiol to dextran to generate a new product with enhanced stability and efficacy. In addition, we have developed methodologies for the synthesis of novel analogues of fused flavonoid natural products, including dependensin, rottlerin and kamalachalcone A. The overall aim of the project is to synthesize novel heterocyclic analogues of isoflavones. The specific objectives of the proposal are to synthesize aza-isoflavone structural analogues and to evaluate their biological activity using in vitro assays to identify structure-activity relationships as a means of refining synthetic targets to ultimately develop lead candidates.
Key Publications
- Falasca V, Wenholz DS, Yu TT, Kumar N. Synthesis of novel isoflavene derivatives with anti-tumour activity. Bioorganic & Medicinal Chemistry Letters 2025, 130350. DOI: 10.1016/j.bmcl.2025.130350
4. Compounds That Activate Glucose Oxidation
Cancer is a major burden of disease, affecting the lives of tens of millions on a global scale. A hallmark feature of nearly all cancer cells is their altered metabolism of glucose compared to non-cancerous cells. Relative to most normal cells, cancer cells use a greater proportion of incoming glucose for non-oxidative purposes including the production of building blocks for cell division (lipid, DNA and protein), rather than oxidative pathways that produce carbon dioxide (CO2) in mitochondria. The goal of this proposal is to develop anticancer molecules that change cancer cell glucose metabolism to be more like that of non-cancerous cells. We have identified a small molecule that increases glucose oxidation and selectively kills cancer cells in vitro and in mice. The aim of this project is to generate new derivatives with enhanced activity and drug-like properties. The new compounds will be evaluated for anticancer activity in various cancer cell lines.
Key Publications
- Byrne FL, Olzomer EM, Marriott GR, Quek LE, Katen A, Su J, Kumar N, Hoehn KL et al. (2020) Phenotypic screen for oxygen consumption rate identifies an anti-cancer naphthoquinone that induces mitochondrial oxidative stress. Redox Biol 28:101374. DOI: 10.1016/j.redox.2019.101374
Environmental Chemistry & PFAS Remediation
Per- and poly-fluoroalkyl substances (PFAS), a class of organofluorine compounds, are attracting intense regulatory scrutiny and public awareness due to their xenobiotic nature and adverse impact to health. PFAS contain extremely stable C-F bonds, have excellent stability, surface activity, oleophobic and hydrophobic properties, and are widely used as water and oil repellents (e.g. in carpets, leather, fire extinguishing agents, non-stick pans, food packaging and other fields). Perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) are the two most common PFAS with half-lives of 5.4 years and 3.8 years, respectively. PFASs are widely used and discharged worldwide, and a variety of PFAS can be detected in the environment, wildlife and in humans. It is now known that PFAS bioaccumulate, biomagnify and are linked to immune suppression and cancer. In this project, we aim to create a platform that combines a sorbent and redox active catalysts (RACs) to remediate PFAS.
Key Publications
- Jun Sun, Sreenu Jennepalli, Matthew Lee, Adele Jones, Denis M. O'Carroll, Michael J. Manefield, Mohan Bhadbhade, Björn Åkermark, Biswanath Das, and Naresh Kumar. Environmental Science & Technology 2022, 56 (12), 7830-7839. DOI: 10.1021/acs.est.1c08254
- Jun Sun, Tsz Tin Yu, Maryam Mirabediny, Matthew Lee, Adele Jones, Denis M. O'Carroll, Michael J. Manefield, Priyank V. Kumar, Russell Pickford, Zeno Rizqi Ramadhan, Saroj Kumar Bhattacharyya, Björn Åkermark, Biswanath Das, Naresh Kumar. Soluble metal porphyrins - Zero-valent zinc system for effective reductive defluorination of branched per and polyfluoroalkyl substances (PFASs). Water Research 2024, 258, 121803. DOI: 10.1016/j.watres.2024.121803
Computational Drug Discovery
1. AI-Driven Drug Design
Amid the growing global AMR crisis, there is an urgent need for efficient methods to discover novel antimicrobial peptides. Integrating computational tools such as virtual screening and mining of genomic data offers a scalable and rapid alternative to traditional approaches. on this collaborative project harnesses advances in computational methods, including machine learning, and deep learning, to enable large scale and accurate screening of AMPs with fewer false positives and the ability to predict activity prior to experimental testing.

2. Molecular Simulation & Dynamics
This project involves the use of molecular docking against key target receptors, integrated with molecular dynamics (MD) and steered MD simulations. This interdisciplinary approach accelerates the drug discovery process and aligns with green chemistry principles by minimizing laboratory waste. It facilitates the development of more effective and selective antimicrobial agents, contributing to the advancement of next-generation therapeutics with enhanced safety and environmental sustainability
Key Publications
- Ibisanmi, T.A., Jiang, X., Willcox, M., Kumar, N. Recent advances in computational antimicrobial peptide discovery through big data, modeling, and artificial intelligence and their interplay in ushering the next golden era of drug development. Front. Bioinform. 2026, 6:1749404. DOI: 10.3389/fbinf.2026.1749404 · PDF
- Naveed Saleem, Naresh Kumar, Emad El-Omar, Mark Willcox, Xiao-Tao Jiang. Harnessing Machine Learning Approaches for the Identification, Characterization, and Optimization of Novel Antimicrobial Peptides. Antibiotics 2025, 14, 1263. DOI: 10.3390/antibiotics14121263
Development of Synthetic Methodologies
Synthesis of Indoles through C2−C3 Bond Formation Using Lawesson's Reagent
We have developed an efficient, metal-free synthetic route for the construction of functionalized indoles through C2–C3 bond formation, mediated by Lawesson's reagent. This versatile methodology employs readily available amidophenylglyoxylic esters and exhibits broad substrate tolerance, yielding a wide range of 2,3-disubstituted indoles. The process is scalable to gram quantities and facilitates diverse downstream modifications, underscoring its significant utility in organic synthesis.
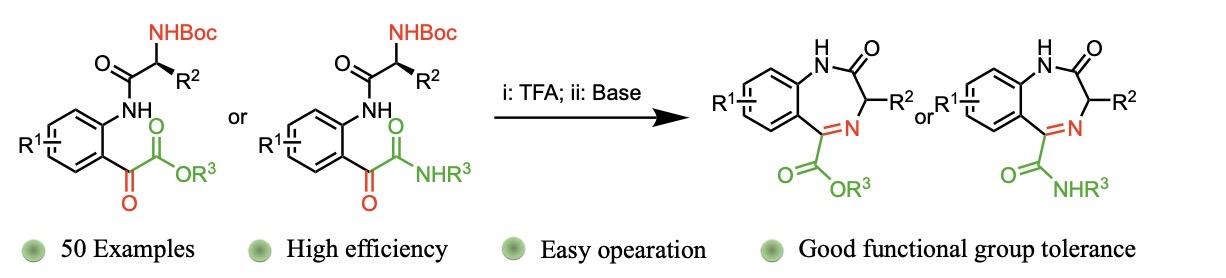
Synthesis of 1,4-benzodiazepin-2-ones from isatins
We have developed an effective synthetic strategy for the construction of 1,4-benzodiazepin-2-ones, a crucial class of compounds in medicinal chemistry with broad therapeutic applications. Our method provides a versatile approach to synthesize derivatives bearing diverse ester or amide functionalities at the C5 position, addressing a previously challenging area. This high-yielding protocol demonstrates excellent substrate tolerance, enabling the efficient preparation of over 50 novel benzodiazepine analogues. Furthermore, the process is scalable to gram quantities, showcasing its potential for practical applications in drug discovery and development.
Key Publications
- Yao Cheng, Tom Yu, Mohan Bhadbhade, David StC Black, Naresh Kumar. Synthesis of Indoles through C2−C3 Bond Formation Using Lawesson's Reagent. J. Org. Chem. 2025, 90, 3290-3300. DOI: 10.1021/acs.joc.4c02922
- Yao Cheng, Thanh Le, Tsz Tin Yu, Mohan Bhadbhade, David StC. Black, and Naresh Kumar. Synthesis of 1,4-Benzodiazepin-2-ones from Isatins. The Journal of Organic Chemistry 2025, 90 (22), 7372-7384. DOI: 10.1021/acs.joc.5c00647
Antimicrobial Strategies for Industrial and Biomedical Applications
1. Tackling Hospital Acquired Infections with Peptide Mimics
The use of medical devices has increased immensely over the last decade. Although, this increase in device use has resulted in a better quality of life and longer patient survival, device-related bacterial infections have emerged as a serious problem with the increased use of medical implants. Once in an established biofilm, bacteria are more resistant to antibiotic treatment and the host immune system. The aim of this project is to develop methodologies for the covalent attachment of novel anti-microbial compounds including specially designed peptide mimics or small heterocyclic compounds onto biomaterial surfaces. Initially the attachment chemistry will be optimised using glass as a model surface, and subsequently the methodology will be adapted for biomaterial surfaces. The resulting biomaterials will be characterised by various surface analysis techniques including XPS and ATR spectroscopy, and assessed for anti-microbial efficacy in collaboration with the microbiology partners.
2. Prevention of Microbial Induced Stress Corrosion Cracking
Microorganisms are known to threaten the longevity of many engineered structures. Previous studies have shown that acidic solutions containing hydrogen sulphide (H2S) can cause hydrogen-induced SCC (HISCC) also known as hydrogen embrittlement (HE). Microorganisms, particularly Sulphate Reducing Bacteria (SRB) present in the mines, produce H2S that results in Microbiologically Induced Stress Corrosion Cracking (MISCC). In this project we aim to develop prevention measures, such as antimicrobial coatings against SRB, as a long-lasting controlling technique to mitigate against MISCC in underground mines.
3. Disinfection Strategies against Microbes and Viruses
The COVID-19 pandemic has occurred through the transmission of the SARS-CoV-2 virus worldwide. All infection control strategies are focussed on reducing the transmission of the virus. Transmission routes for SARS-CoV-2 are through aerosols of respiratory secretions and via contamination of surfaces. Whilst there are currently several vaccines that prevent a person becoming seriously ill with the virus and reduce the load of virus within individuals, there is still uncertainty on how quickly they can be deployed to control the spread of the disease around the world. This means strategies for reducing transmission of the virus may well be in place for many years to come. The main aim of this project is to develop disinfection strategies to decontaminate surfaces and equipment for reuse or recycling.
4. Development of Novel Environmentally Benign Technologies for the Control of Biofilms
There is an imperative to develop environmentally compatible strategies to control bacterial biofilms on industrial surfaces. For example, biofilm mediated corrosion affects a range of industries, from oil distribution to food processing surfaces and has been estimated to result in added costs of between US$20–300 billion per year in the USA alone. Biofilms also form on membrane surfaces, such as in reverse osmosis plants, where the associated biomass block the membrane pores. Fouling increases the water pressure required to continue filtration, significantly increasing the cost of water purification. When fouling occurs, water purification systems must be shut-down for chemical disinfection and back flushing to alleviate fouling. The current state of the art for the removal of biofilms from pipelines, membranes and food handling surfaces typically include either alone or in combination, mechanical scrubbing, such as 'pigs' or brushes as well as harsh chemicals, many of which are highly toxic, such as glutaraldehyde. These low-tech approaches are typically not effective at removing biofilms which are significantly more resistant than free living bacteria.
Recently, it has been shown that by exogenously adding small molecules that generate nitric oxide (NO), generically called NO donors, it is possible to induce the dispersal of surface associated bacteria and biofilms in a number of bacteria. The aim of this project is to deliver nitric oxide (NO) donating molecules directly on surfaces using coatings or co-polymers for the control of biofilms on surfaces.